
Liver fibrosis is a pathophysiological process, which describes the abnormal proliferation of connective tissues in liver triggered by various pathogenic factorsIn all cases of liver damage, liver fibrosis will be triggered during liver repair and healing. If the damaging factor cannot be eliminated in the long term, liver fibrosis would persist and eventually develop into liver cirrhosis.
Type-III serum procollagen N-terminal peptide is an N-terminal propeptide released into the blood circulation when extracellular type-III procollagen is converted into type-III collagen by specific enzymes.Its amount is equivalent to type-III collagen. Therefore, assessing the amount of type-III serum procollagen N-terminal peptide allows the evaluation of the amount and rate of type-III collagen synthesis. Because type-III collagen primarily exists in the liver, the amount of type-III serum procollagen N-terminal peptide can reflect the activity of liver fibrosis and other liver diseases.Assessing its amount helps distinguish mild hepatitis versus moderate/severe chronic hepatitis, and indicate the existence of active liver fibrosis.The assessment results can serve as an indicator of the activity and severity of chronic liver fibrosis.
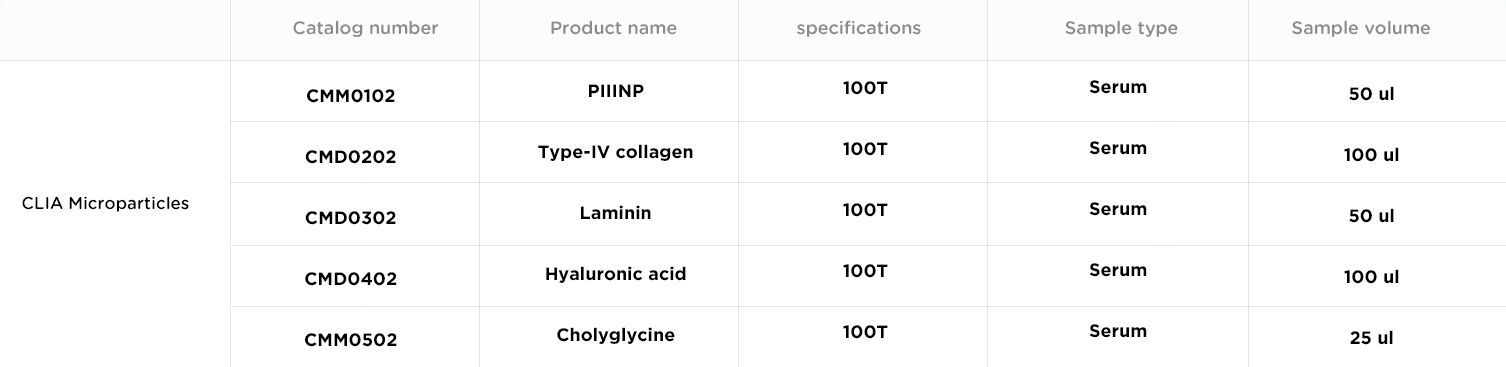
Type-IV collagen is a major component of the basement membrane.The Disse space of health liver tissues does not contain basement membrane, and therefore only has an extremely low amount of type-IV collagen. However, during the progression of hepatitis towards liver fibrosis and further towards liver cirrhosis, basement membrane will be formed in the Disse space.As a result, the amount of type-IV collagen in liver tissues and blood will also increase.
Laminin (LN) is a macromolecular glycoprotein that is mainly produced in adipocytes, hepatocytes and endothelial cells.Its main function is to form the basement membrane zone together with other molecules like type-IV collagen, and to maintain its stability. During the progression of liver fibrosis and liver cirrhosis, the synthesis of LN is up-regulated, and the generated LN is deposited into the Disse space. As shown by previous studies, the weaker the liver function in liver cirrhosis patients, the higher their serum indexes.Therefore, serum indexes can be used to indicate the capillarization of hepatic sinusoids and the fibrosis of interlobular portal areas.
Hyaluronic acid (HA) is widely distributed in the extracellular space.It is primarily synthesized in fibroblasts in tissues, and enters blood circulation through the lymphatic system, and is rapidly cleared by liver. Many liver diseases, which can be caused by various factors, can induce the up-regulation of serum HA level, mainly due to the weakened clearance function of liver and the increase of liver fibroblast (hepatic stellate cell) production. During liver fibrosis, the capacity of hepatic stellate cells in synthesizing HA is greatly elevated;meanwhile, the capacity of endothelial cells in clearing HA is weakened.These two factors cooperatively induce the up-regulation of HA level in liver fibrosis.
Serum cholyglycine (CG) is a conjugated bile acid that is generated by the conjugation of cholic acid with glycine. It is a major component of bile acids. When damage occurs in liver cells, the capability of liver cells to ingest CG is down-regulated, which results in the elevation of CG level in the blood. During cholestasis, the liver becomes deficient in excreting cholic acid.The amount of CG in the reflux blood circulation becomes higher, which also induces the elevation of CG level in the blood. Thus, assessing the serum CG (SCG) level provides a sensitive indicator of liver cell functions and also the substance circulatory function of the hepatobiliary system.
[1] Annoni G, Cargnel A, Colombo M, Hahn EG (1986) Persistent elevation of the aminoterminal peptide of procollagen type III in serum of patients with acute viral hepatitis distinguishes chronic active hepatitis from resolving or chronic persistent hepatitis. J Hepatol 2:379–388.
[2] Khoshnoodi J, Pedchenko V, Hudson BG (May 2008). "Mammalian collagen IV". Microsc. Res. Tech. 71 (5): 357–70.
[3] Timpl R et al. (1979). "Laminin – a glycoprotein from basement membranes". J Biol Chem 254 (19): 9933–7.
[4] Yurchenko P and Batton BL (2009). "Developmental and Pathogenic Mechanisms of Basement Membrane Assembly". Curr Pharm Des. 15 (12): 1277–94.
[5] Hyaluronate Sodium in the ChemIDplus database, consulté le 12 février 2009.
Address: NO.87 Jingbei Yi Rd, National Eco&Tech Zone, Zhengzhou, China
Email: info@autobio-diagnostics.com Tel: +86-371-6200-7036
Autobio Copyright Reserved for ICP 18006568. All Rights Reserved.




